What’s the Future of Patient Centricity?
 5 years ago
By Josh Neil
5 years ago
By Josh Neil
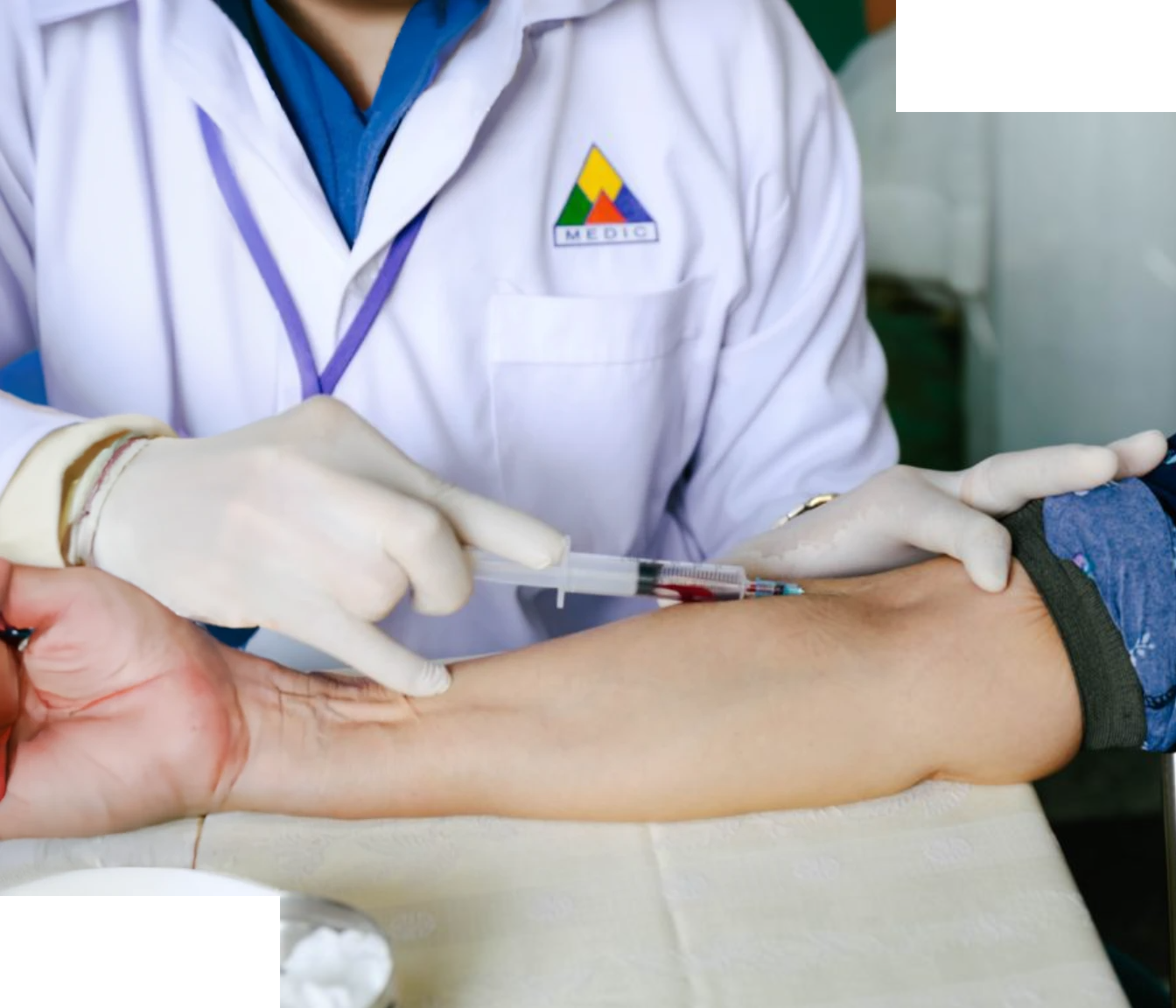
Clinical trials rely fundamentally on patient engagement. While these trials can often prove life-saving, patient recruitment and retention has long been an issue for researchers. Simply increasing the amount of patients in a study means delays, increased costs, and patients receiving medicines more slowly. Traditional methods for bringing patients into trials are not enough. As companies have realised this, patient centricity has become more and more popular.
Patient centricity is the answer to many of the problems facing clinical trials. It ensures clinical decisions focus largely on the patient. But this addition to clinical trials is still an uncertain concept for many, and its possible application even more so. We decided to look into the burgeoning need for patient centricity, and its application in the next few years.
What is Patient Centricity?
There are few now in the pharma industry who don’t understand the concept of patient centricity. The official designation describes it as any decision or solution which puts the patient first. This involves preferably collaborating with the patient on that decision, and understanding their needs before action occurs.
It’s certainly a necessary step forward in pharmaceuticals, and one many companies have incorporated both into their practices and ethos. But it’s still a vague definition, one which could be interpreted in numerous ways by different companies.
Fundamentally, patient-centrism should be a fundamental discourse with the patient on any decision involving them. A company’s values full incorporate the practice if clinicians make no protocol without patient agreement. Naturally, stakeholders will still hold the most important decision-making power overall, but patients will also play a key role.
How Companies are Working with Patient Centricity
There are a number of simple, easily effectable steps organisations can make to ensure greater patient centricity in their studies. These include:
- Paying attention to the content and style of the patient information document—when writing this, it is important to see it as a way to inform patients of their participation in a trial, and to help them understand how their conditions will impact the trial, the visit schedules for the study and any potential risks they face
- Ensure patients have a say in the design and endpoint designation of the trial—while it is certainly important to ensure clinical endpoints are focused around regulatory guidelines and symptom severity, patients must also be allowed to decide what is important to them. Patient preference studies can help determine the balance between regulatory-relevant endpoints and patient needs
- Regular updates should be given to the patient to ensure they are informed about the specifics of the study, the value of their information and any changes around timeline that occur
Where is Patient Centricity Going Next?
This focus on interaction with the patient is driving the industry to bring in trial populations earlier and earlier. This helps researchers develop programs which solve patient issues as an integral part of their being, not as an afterthought. This can have ramifications for trial efficacy, with greater improvements expected in trials which put patients foremost during design.
This change is also seeing a wider involvement of stakeholders around patient wellbeing. Involving site staff in program creation can ensure greater understanding of bottlenecks and cost issues. This also allows delivery of medicine direct to the patient. Patient advocacy groups are also influential in trial design, with their expertise and direct communication with patients.
For the most part, companies must in the near future simply expand practices that many are already implementing. This means engaging more not only with patients but with advocacy groups and stakeholders, and supporting better outcomes for trial patients.
As pharma companies progress they will also naturally come to understand the perfect balance between different outcomes, as demanded by payers, patients and clinicians, that can best support patient centricity in a pragmatic way. While not all outcomes will be totally satisfactory to every party, with enough experience companies can balance requirements to keep payers happy while ensuring patients get the best experience possible.
Despite the constant move towards patient centricity, some challenges do remain. Much has been made of the dangers of direct-to-consumer marketing, and its potential for causing harm and misinformation in the public space. Current regulatory compliance has been criticised for preventing pharma companies from correcting unregulated sources of information, most famously in the case of the MMR vaccine and the ‘anti-vax’ movement.
Technology and Data
Technology is naturally another area in which companies are exploring the depths of increased patient centricity. The mobile health market has grown precipitously in recent years, with 2020 set to see greater take-up of technologies that will allow patients not only to share their data with clinicians but also see more information on their trial, treatments, and healthcare checkpoints along the way.
The statistics back up this viewpoint. In 2020, around 40% of all Internet of Things devices are healthcare-related. This change has a significant impact on patients: another study found that 53% of patients would prefer a provider supplying remote or telemonitoring devices.
A solution that will have significant impact in 2020 and beyond is the interoperability platform. Unstructured data has long been a particular problem in the clinic, where doctors’ notes, images and various forms have created a struggle for automated systems to compile and save them in any meaningful way. With the slow push to structure all clinical data, the interoperability platform will have more and more of an impact. This central system will allow various sources of patient data to be compiled, securely held and shared privately within the ecosystem of the trial.
AI will also, naturally, improve selection and recruitment of patients and recruited for clinical trials. The means that to use social media and other online resources to identify sub-populations and better stratify potential patients is a huge boon for clinicians, saving them large amounts of time and money in selecting patients and inevitably dealing with retention issues and other external factors. AI will find better, more selective patients, quicker and more easily than before.
The benefits AI and technology will bring, and the bonus to patient selection and retention that follow, cannot be ignored. Patient centricity is hugely beneficial for both clinical trials and for the patients themselves. It can save pharma companies and researchers large amounts of time and money. Companies must act on this trend as possible to reap those benefits, as the pharma industry brings the patient more than ever into the conversation.
Joshua Neil, Editor
Proventa International
To ensure you remain up-to-date on the latest in clinical development, sign up for Proventa International’s Clinical Operations, Supply Chain & Pharmacovigilance Strategy Meeting 2020, hosted on 11 May in San Diego, USA.

How to Reduce the Cost of Clinical Trial Supply Chain While Maintaining Quality and Efficiency
Clinical trial supply chain is a critical component of drug development, ensuring that drugs are available for testing, regulatory approval, and distribution. However, managing this supply chain can be challenging, with factors such as cost, quality, and efficiency all coming...
2 years agoHow to Reduce the Cost of Clinical Trial Supply Chain While Maintaining Quality and Efficiency
Clinical trial supply chain is a critical component of drug development, ensuring that drugs are available for testing, regulatory approval, and distribution. However, managing this supply chain can be challenging, with factors such as cost, quality, and efficiency all coming...
2 years ago
Raising Representation in Clinical Trials: An Interview with Liz Beatty, Inato
The COVID-19 pandemic created a significant shortage of suitable patients for clinical trials worldwide. Within this crisis was another more serious issue around inclusion and representation, driven by COVID’s disparate effect on certain ethnicities, that spoke to a wider issue...
4 years agoRaising Representation in Clinical Trials: An Interview with Liz Beatty, Inato
The COVID-19 pandemic created a significant shortage of suitable patients for clinical trials worldwide. Within this crisis was another more serious issue around inclusion and representation, driven by COVID’s disparate effect on certain ethnicities, that spoke to a wider issue...
4 years ago
DeepMind’s AI Predicts Structures for More Than 350,000 Proteins
In 2003, researchers sequenced approximately 92% of the human genome, a huge achievement and very recently researchers have completed the entire process. Now, the latest innovation in AI technology has predicted the structure of nearly the entire human proteome. The...
4 years agoDeepMind’s AI Predicts Structures for More Than 350,000 Proteins
In 2003, researchers sequenced approximately 92% of the human genome, a huge achievement and very recently researchers have completed the entire process. Now, the latest innovation in AI technology has predicted the structure of nearly the entire human proteome. The...
4 years ago